Exploration of Protein Markers in Lyme Disease
by Rachel Zhang
Background
Lyme disease is a debilitating disease that attacks all over the body, namely the joints and muscle, caused by the bacteria Borrelia burgdorferi. It is spread by the Ixodes tick, deer tick, and is endemic to the Northeastern United States. There are 100,000 new cases in the United States every year.
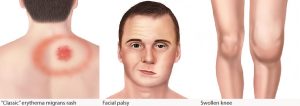
The first symptom is a bull’s-eye rash around the tick bite. Fever, fatigue, joint pain and muscle pain usually follow within three to thirty days. Facial paralysis, arthritis and heart disease will appear in the late stages of the disease, days or even months after infection.
Borrelia burgdorferi, the bacteria, is a bacterial species of the spirochete class of the genus Borrelia. Borrelia species are considered to be diderm (double-membrane) bacteria.
Current Treatment
Lyme disease is treated by oral antibiotics such as doxycycline or amoxicillin or intravenous ceftriaxone. Antibiotic treatment is halted after a month as there is no evidence that further treatment is effective.
The Problem
10-20% of Lyme disease cases develop into Chronic Lyme disease. Chronic Lyme disease, also known as post-treatment Lyme disease syndrome appears in patients who have been already treated for acute Lyme disease. The medical community, however, is unsure as to the true underlying cause of Chronic Lyme disease. Currently there is no evidence that the residual pain that Lyme disease patients suffer through after treatment is the result of continued infection. The two theories are that Chronic Lyme disease is the result of Borrelia burgdorferi bacteria that haven’t been isolated and treated, or that the symptoms are the result of the damage caused to nerves during initial infection. Evidence currently shows that patients with acute Lyme disease have clear differences in blood protein composition than that of uninfected controls. In addition, patients with Chronic Lyme disease have no significant difference in blood protein composition than that of uninfected controls, which strongly suggests that no bacteria is present.
Project
My project was to optimize urine protein enrichment procedure for investigating Borrelia surface proteins in Lyme patient urine in order to be able to clearly study the gel. This contributes to the bigger project of whether Borrelia burgdorferi is the cause of Chronic Lyme disease. In 1991, the detection of Borrelia burgdorferi in urine was shown by Goodman. Since the Borrelia protein concentrations in urine are too low to be detected by the detection method, we need to enrich the samples first.
Results
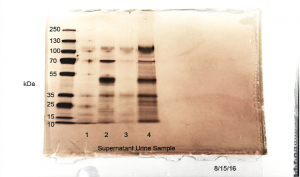
As shown in Figure 1, proteins in all four urine samples processed in this experiment were successfully concentrated without significant signs of degradation (typical bands or smear in < 10KDa MW area) or precipitation (smear in top >130 KDa area).
In conclusion, the ProteoSpin Urine Protein Concentration Micro Kit provides a simple and straightforward method to concentrate urine proteins with minimum degradation, sample loss and precipitation. The dilution ratio we discovered provides the clearest gel pictures. This same procedure is also able to simultaneously remove the smell, color and solvents from the urine, which is very helpful for downstream sample analysis.
These processed urine samples have been further analyzed in the lab by mass spectrometry and other antibody-based tests. The same urine protein concentration procedure is going to be applied in ongoing large-scale clinical sample analysis for Lyme disease biomarker discovery.
Figure 1, Silver staining SDS-PAGE gel showing urine proteins after concentration. All four urine samples processed in this experiment presented positive protein bands in MW range from 10KDa to 120 KDa.