Lipid Accumulation Under Phosphate Deprivation
by Christine D. Pham
Experiment
Global warming resulting in climate change is pressing issue that this generation faces. The burning of fossil fuels and release of carbon dioxide into the atmosphere is a major contributor to global warming. With fossil fuels being a finite resource, there is an urgent need for an alternative sustainable energy source to petroleum and natural gas. An alternative to fossil fuels: lipid-producing microalgae biofuel is more environmentally sustainable and can be grown in large quantities. Chlamydomonas reinhartii is a type of microalgae commonly used in biofuel research. The lipid primarily produced by C. reinhartii is triacylglycerol, the same fatty acid which are used to produce biodiesel.
Experiment
As part of my internship at ISB, I teamed up with Jordan Raymond, an undergrad intern, to quantitatively determine the rate at which lipid accumulation in Chlamydomonas reinhartii occurs under phosphate deprivation. Nitrogen and phosphate are essential nutrients for the cell growth and division of C. reinhardtii. When one is absent, cell division ceases and lipid accumulation begins. This experiment was conducted in photobioreactors; a population of C. reinhartii CC-1690 in a TP-NO3 bioreactor. TP-NO3 media is complete with all the nutrients to necessary to grow C. reinhardtii.

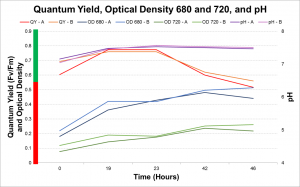
Daily measurements (i.e. Quantum yield, Optical Density at 680nm, Optical Density at 720nm, pH) were taken until a high cell count was achieved. These healthy cells, based on a high quantum yield, or photosynthetic efficiency, were then spun down and washed with phosphate deprived media. The concentrated cells were then suspended in phosphate deprived media and then inoculated in T-NO3 bioreactors.
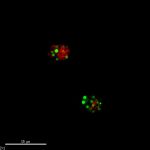
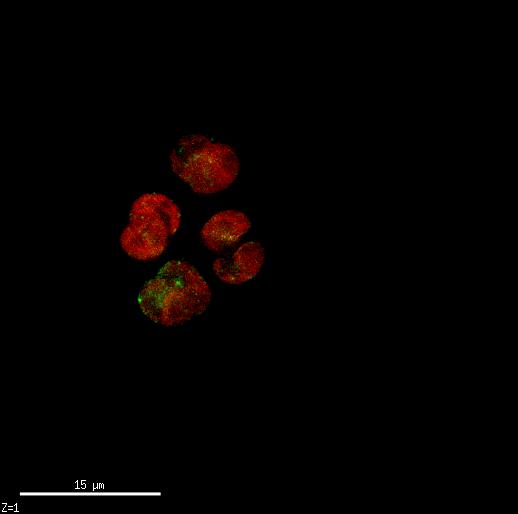
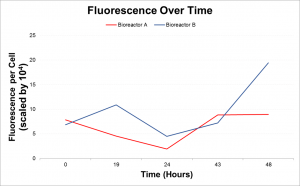
The lipid accumulation was recorded twice daily using Nile Red. Lipid accumulation can be recorded via a 96-well plate reader which emits light, exciting the cells at approximately 500nm and then recording the emission at approximately 575nm. These readings get normalized to the cell count to calculate Fluorescence per cell. Lipid accumulation can also be seen in cells with an epi-fluorescence microscope where the cells glow red and the lipid inside glows bright yellow.
The phosphate limitation experiment was ran twice. The first run did not produce lipids as rapidly as hoped. Upon inspecting Nile Red stained cells under an epifluorescent microscope, it was evident that some cells were accumulating lipids while others were not.
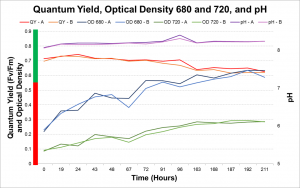
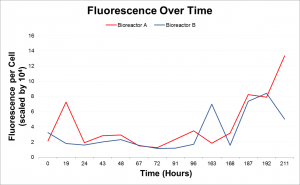
As a result it was determined that more intense light and a higher concentration of CO2 would increase the rate of lipid accumulation. In the second run the photobioreactors were moved closer to the fluorescence light and the concentration of CO2 pumped into the photobioreactors was increased. The second run of the experiment showed that with increased light and CO2 the rate of lipid accumulation with phosphate deprivation is significantly faster.
Conclusion
After intense light exposure and increased carbon dioxide, a phosphate limitation can rapidly increase lipid accumulation in Chlamydomonas reinhartii. This serves as groundwork for potential mass production of biofuels through limited nutrients to promote lipid accumulation rather than cell growth.
