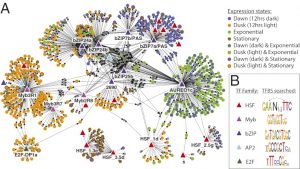
Marine diatoms are important primary producers that thrive in diverse and dynamic environments. Using the model species Thalassiosira pseudonana, we demonstrated in a detailed physiological and transcriptomic survey that approximately 40 percent of the transcriptome varied significantly and recurrently, reflecting large, reproducible cell-state transitions between four principal states: I) "dawn," following twelve hours of darkness, II) "dusk," following twelve hours of light, III) exponential growth and nutrient replete, IV) stationary phase and nutrient depleted. Repeated shifts in the transcript levels of hundreds of genes encoding sensory, signaling, and regulatory functions accompanied the four cell-state transitions, provided a preliminary map of the highly coordinated gene regulatory program under varying conditions. These results explain, in comprehensive detail, how the diatom gene regulatory program operates under varying environmental conditions (Ashworth et al. 2013). Furthermore, we have shown than T. pseudonana rapidly responds to increasing CO2 by differentially expressing gene clusters that regulate transcription and chromosome folding, then reduces expression of photosynthesis causing a shift in regulation and metabolism re-arrangement (Hennon et al. 2015). Furthermore we recently demonstrated that diatoms are more sensitive to collapse when at low carbon and facing multiple perturbations suggest that ocean acidification will stabilize diatom populations with a potential to see an increase in their niche broadening (Valenzuela et al. 2017). Additionally we have integrated all existing publicly available microarray data for the diatom models T. pseudonana and Phaeodactylum tricornutum online, and explorable through the Diatom Portal (Ashworth et al. 2015). This detailed knowledge of this dynamic molecular process is invaluable for new hypothesis generation and the interpretation of genetic, environmental and meta-transcriptomic data from field studies.
We also successfully integrated interdisciplinary research and education efforts. We developed an inquiry-based high school education module to teach the process of systems biology in context of climate change, carbon cycling and ocean acidification using T. pseudonana as a model system (Ludwig et al. 2015). Using via Google Analytics we have track as many as 160,000 students completing our curriculum each year from 39 states and numerous countries.
